TYO:6869
Sysmex Corporation commands over 50% of the global hematology analyzer market, processes billions of blood tests annually across 190+ countries, and is now expanding aggressively into cancer precision medicine through liquid biopsy technology. This deep-dive report examines the Kobe-based diagnostics giant’s financial performance, product portfolio, regional strategy, and partnership opportunities for international businesses.
Executive Summary
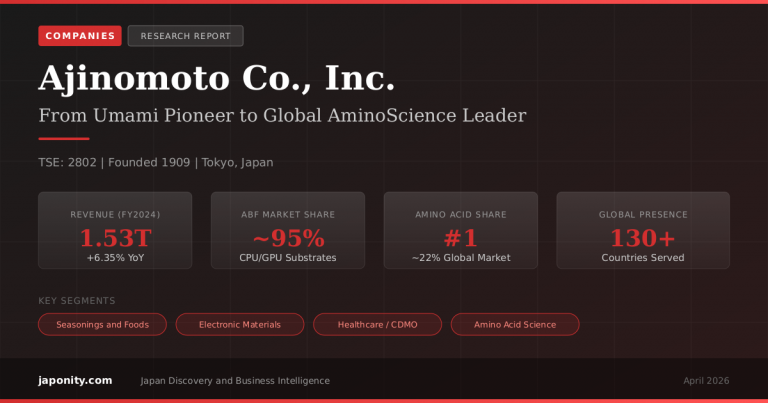
Sysmex Corporation (TYO: 6869) is the undisputed global leader in hematology diagnostics, holding an estimated 50–60% share of the worldwide blood cell analyzer market. Founded in 1968 in Kobe, Japan, with just eight employees, the company has grown into a ¥508.6 billion (US$3.4 billion) revenue enterprise with 11,457 employees, 79 global subsidiaries, and a presence in over 190 countries. Beyond its dominance in hematology, Sysmex is strategically expanding into hemostasis, urinalysis, clinical chemistry (through its acquisition of JEOL‘s medical equipment business), and cancer precision medicine via its subsidiary Sysmex Inostics.
Company Overview
| Item | Details |
|---|---|
| Official Name | Sysmex Corporation (シスメックス株式会社) |
| Founded | 1968 (as TOA Medical Electronics) |
| Renamed | 1998 (to Sysmex Corporation) |
| Headquarters | Kobe, Hyogo, Japan |
| Stock Listing | Tokyo Stock Exchange Prime (6869) |
| Employees | 11,457 consolidated (as of March 2025) |
| Global Presence | 190+ countries, 79 subsidiaries |
| FY2025 Revenue | ¥508.6 billion (US$3.4 billion) |
| Core Business | In vitro diagnostics (IVD) |
Corporate History
Sysmex’s journey began in 1968 as a division of TOA Corporation focused on medical electronics. The company developed Japan’s first automated hematology analyzer, a milestone that set the foundation for its global dominance. It was formally established as an independent entity in 1978 and rebranded as Sysmex Corporation in 1998 to reflect its expanding global identity. Over the decades, it has consistently invested in R&D to push the boundaries of automated diagnostics, moving from simple blood cell counting to AI-powered multi-parameter analysis.
Hematology Dominance
Sysmex’s position in the hematology market is extraordinary. With an estimated 50–60% global market share in hematology analyzers and reagents—up from approximately 40% a decade ago—the company is the single largest player in a market valued at US$4.33 billion in 2025 and projected to reach US$7.28 billion by 2034.
What Makes Sysmex Dominant
- XN-Series platform — The flagship automated hematology analyzer line, including the high-volume XN-1000/XN-2000 and compact XN-350, serves both large reference laboratories and point-of-care settings
- XR-Series — The next-generation platform featuring advanced fluorescence flow cytometry and AI-enhanced digital morphology
- Razor-and-blade model — Instruments generate recurring reagent revenue, creating a highly predictable revenue stream that competitors struggle to displace
- AI integration — AI-powered complete blood morphology (CBM) analysis combines CBC data with deep learning algorithms trained on millions of patient samples
- 40+ years of clinical validation — Decades of peer-reviewed evidence and installed base create enormous switching costs
Hematology Market Landscape
| Company | Estimated Share | Key Platform | Strength |
|---|---|---|---|
| Sysmex | 50–60% | XN-Series, XR-Series | Market leader, AI morphology, deepest reagent menu |
| Beckman Coulter (Danaher) | ~15% | DxH 900 | Impedance technology, integrated lab automation |
| Abbott | ~10% | CELL-DYN Sapphire | Broad diagnostics portfolio, Alinity integration |
| Siemens Healthineers | ~8% | ADVIA 2120i | Integrated workflow with chemistry/immunoassay |
| Others | ~10–15% | Various | Regional players, emerging market brands |
In fiscal Q1 2025, Sysmex posted a remarkable 19.2% growth in hematology sales, driven by the adoption of AI-enhanced analyzers globally.
Product Portfolio
Sysmex has strategically diversified beyond hematology while maintaining its core focus on laboratory diagnostics:
| Business Area | Key Products | Market Position | Recent Milestones |
|---|---|---|---|
| Hematology | XN-Series, XR-Series, XN-L Series | #1 globally (50–60%) | AI-powered digital morphology |
| Hemostasis | CN-6000, CN-3000, CN-9000 system | #1 globally | FDA clearance for CN-6000 (June 2025) |
| Urinalysis | UF-Series, UC-Series | #1 globally | Integrated with XN platform |
| Clinical Chemistry | BioMajesty (via JEOL acquisition) | Entering market | Acquisition closing April 2026 |
| Flow Cytometry | Research & clinical analyzers | Growing niche | Sysmex Partec integration |
| Life Science | OncoBEAM, Plasma-SeqSensei | Emerging leader | LabCorp partnership for oncology |
CN-Series: The Hemostasis Breakthrough
Sysmex’s CN-9000 modular hemostasis solution represents a significant competitive advance. Supporting up to three CN-3000 or CN-6000 analyzers, the system processes up to 1,000 samples per hour and leverages universal components shared with the XN-Series hematology and UF-Series urinalysis platforms. The June 2025 FDA clearance of the CN-6000 opens a major growth vector in the U.S. market.
JEOL Acquisition: Entering Clinical Chemistry
In September 2025, Sysmex announced the acquisition of JEOL’s clinical chemistry testing business, expected to close in April 2026. JEOL’s BioMajesty analyzer is known for minimal sample volumes and reduced reagent consumption, and holds a strong market share in Japan. By combining this with Sysmex’s global sales network, the company gains access to the large clinical chemistry market—particularly in emerging countries comprising 85% of the world’s population where basic screening tests are still being adopted.
Financial Analysis
Revenue Trajectory
| Metric | FY2023 | FY2024 | FY2025 | H1 FY2026 |
|---|---|---|---|---|
| Revenue (¥ Bn) | 427.4 | 471.4 | 508.6 | 232.5 |
| Revenue (US$ Bn) | 2.9 | 3.2 | 3.4 | 1.6 |
| Operating Profit (¥ Bn) | 72.8 | 77.5 | 82.1 | 32.9 |
| Net Income (¥ Bn) | 54.2 | 56.0 | 60.3 | 19.0 |
H1 FY2026 Challenges
The first half of FY2026 (April–September 2025) showed a temporary setback: revenue declined 4.1% year-on-year to ¥232.5 billion, while operating profit fell 25.9% to ¥32.9 billion. Three factors drove the decline:
- Japan system transition — A major IT system migration restricted order processing in Q1
- Yen appreciation — Currency headwinds reduced the value of overseas earnings
- China market softness — Revenue from China declined to 79.4% of prior-year levels amid regulatory and procurement changes
However, the company views these as transitory issues. Recovery is underway, with strong performance in EMEA (+5.6%), the Americas (+1.5%), and Asia Pacific (+5.7%) partially offsetting the China weakness.
Business Model Strength: Reagent Recurring Revenue
A key feature of Sysmex’s financials is the razor-and-blade business model. Instrument placements generate long-term reagent revenue, which accounts for the majority of total sales. Each installed analyzer creates a multi-year revenue stream that is highly resistant to competitive displacement. This model provides predictability, high gross margins, and strong cash flow generation.
Regional Strategy
Revenue by Region (H1 FY2026)
| Region | Revenue (¥ Bn) | Share of Total | YoY Change | Strategic Priority |
|---|---|---|---|---|
| EMEA | 73.0 | 31.4% | +5.6% | Hemostasis expansion, liquid biopsy commercialization |
| Americas | 64.7 | 27.8% | +1.5% | CN-6000 launch, Sysmex Inostics oncology testing |
| China | 43.3 | 18.7% | -20.6% | Market recovery, localized production |
| Asia Pacific | 26.2 | 11.2% | +5.7% | Emerging market penetration, basic screening |
| Japan | 25.4 | 10.9% | -3.8% | System transition recovery, clinical chemistry entry |
Regional Highlights
- EMEA (31.4%) — The largest region by revenue. Hemostasis has been a particular growth driver in Europe, and Sysmex is commercializing Plasma-SeqSensei liquid biopsy kits across the EMEA region
- Americas (27.8%) — The second-largest market is positioned for acceleration with the CN-6000 FDA clearance and expanding oncology partnerships through Sysmex Inostics and LabCorp
- China (18.7%) — Despite the 20.6% decline, China remains a strategically critical market. Sysmex anticipates recovery as procurement normalization progresses
- Asia Pacific (11.2%) — The fastest-growing region, driven by hospital infrastructure investment across Southeast Asia, India, and Oceania
- Japan (10.9%) — The home market is expected to recover as the system transition completes and the JEOL clinical chemistry business adds a new revenue layer
Cancer & Precision Medicine
Sysmex’s most transformative growth opportunity lies in its oncology and precision medicine business, led by subsidiary Sysmex Inostics based in Baltimore, Maryland.
Liquid Biopsy Technology
Liquid biopsy—the analysis of cell-free tumor DNA (ctDNA) from blood samples—is one of the fastest-growing segments in diagnostics, with the global market expected to grow at a CAGR of 16.6% through 2032. Sysmex’s key technologies include:
- OncoBEAM — Digital PCR-based platform for ultra-sensitive detection of tumor mutations in blood
- SafeSEQ — Ultra-sensitive next-generation sequencing technology for early-stage cancer detection and monitoring
- Plasma-SeqSensei Breast Cancer IVD Kit — Commercially launched in EMEA for blood-based breast cancer mutation profiling
- Plasma-SeqSensei Solid Cancer IVD Kit — Broader solid tumor panel launched in EMEA
Strategic Partnerships in Oncology
| Partner | Collaboration Focus | Significance |
|---|---|---|
| LabCorp | Blood-based molecular testing services | Access to LabCorp’s global central lab network for precision medicine commercialization |
| Genomic Testing Cooperative | Tissue & liquid biopsy assay services | Covering solid tumor, hematology, and liquid biopsy profiles |
| Clearbridge BioMedics | Circulating tumor cell (CTC) detection | Joint development of new liquid biopsy systems |
The liquid biopsy market is still in its early commercialization phase, but Sysmex is positioned as one of the top five global players. As regulatory approvals expand and clinical evidence accumulates, this segment could become a significant revenue contributor within 5–10 years.
Competitive Landscape
| Company | HQ | IVD Revenue (US$ Bn) | Hematology Strength | Differentiation |
|---|---|---|---|---|
| Sysmex | Japan | ~3.4 | #1 (50–60% share) | Hematology focus, AI morphology, liquid biopsy |
| Roche Diagnostics | Switzerland | ~17.0 | Minor | Broadest IVD portfolio, immunoassay leader |
| Abbott Diagnostics | USA | ~12.5 | ~10% share | Alinity integration, rapid testing, POC |
| Beckman Coulter (Danaher) | USA | ~8.5 | ~15% share | Lab automation, clinical chemistry breadth |
| Siemens Healthineers | Germany | ~6.0 | ~8% share | Integrated imaging + diagnostics |
Sysmex’s Competitive Advantages
- Category dominance — No other company comes close to Sysmex’s hematology market share. This creates massive scale advantages in R&D amortization, reagent manufacturing, and service network density
- Installed base moat — Hundreds of thousands of analyzers globally create a locked-in reagent revenue stream. Switching costs are prohibitive for hospitals due to retraining, revalidation, and workflow integration
- Three #1 positions — Sysmex holds the top global market share in hematology, urinalysis, and hemostasis—a unique trifecta in the IVD industry
- R&D-to-market speed — As a diagnostics-focused pure play, Sysmex can invest more efficiently than diversified conglomerates like Roche or Abbott
- Emerging market opportunity — The JEOL acquisition positions Sysmex to offer a complete basic screening package (hematology + chemistry + urinalysis) to the 85% of the world’s population where these tests are still being adopted
Key Risks
- China market uncertainty — Regulatory and procurement changes have already impacted H1 FY2026 results, and geopolitical tensions add further risk
- Currency exposure — With nearly 90% of revenue from overseas, yen fluctuations significantly impact consolidated results
- Concentration risk — Heavy reliance on hematology means any disruption to this segment would disproportionately impact the company
- Liquid biopsy competition — Well-funded competitors like Guardant Health, Foundation Medicine (Roche), and Grail are also aggressively pursuing liquid biopsy markets
Business Opportunities
For international companies seeking to engage with Sysmex or leverage its market position, several strategic avenues exist:
For Healthcare & Laboratory Partners
- Reagent co-development — Sysmex’s open platform approach creates opportunities for specialized reagent manufacturers to develop compatible assays
- Laboratory automation integration — System integrators can partner with Sysmex to build end-to-end laboratory workflows combining hematology, hemostasis, and chemistry
- Emerging market distribution — As Sysmex expands into markets where basic screening is still growing, local distributors and service partners are in demand
For Technology Companies
- AI and digital health — Sysmex is actively integrating AI into its analyzer platforms; startups with machine learning, computer vision, or clinical decision support capabilities are potential partners
- Liquid biopsy analytics — Bioinformatics companies specializing in ctDNA analysis, variant calling, or multi-cancer early detection could find synergies with Sysmex Inostics
- Cloud-based laboratory informatics — Sysmex’s Caresphere informatics platform could benefit from partnerships with cloud infrastructure and cybersecurity providers
For Investors
- Recurring revenue resilience — The reagent-driven business model provides defensive characteristics even during economic downturns
- Multiple growth vectors — Hemostasis expansion, clinical chemistry entry, and liquid biopsy commercialization provide layered growth optionality
- Emerging market exposure — Unlike peers that depend heavily on developed markets, Sysmex has significant growth runway in underserved populations
Outlook
Sysmex Corporation stands as a rare example of a Japanese company that has achieved and sustained global category dominance. Its 50–60% share of the hematology analyzer market is a competitive position unmatched in most segments of the diagnostics industry, and the company continues to extend its lead through AI-powered innovation and platform breadth.
Looking forward, the company’s growth narrative rests on three pillars. First, the hemostasis business is experiencing rapid adoption in Europe and the United States, bolstered by the CN-6000 FDA clearance. Second, the JEOL acquisition brings clinical chemistry capabilities that complete Sysmex’s basic screening portfolio and unlock significant emerging market opportunity. Third, the liquid biopsy business, while still early stage, positions Sysmex at the frontier of cancer precision medicine—a market growing at 16%+ annually.
The near-term headwinds from China and currency effects are real but manageable within the context of a globally diversified, recurring-revenue business. Sysmex’s forward guidance anticipates strong revenue and profit recovery in FY2027 and beyond, driven by overseas momentum and new product launches.
For global businesses, Sysmex represents a compelling engagement opportunity—whether as a technology partner, distribution collaborator, or investment target. Its combination of market leadership, innovation pipeline, and geographic diversification makes it one of the most strategically important companies in the global healthcare industry.
Need deeper analysis on Sysmex or other Japanese companies?
Japonity provides custom research reports, market entry analysis, and partner matching services for businesses targeting the Japanese market. Contact us to discuss your needs.
Interested in Japanese business opportunities?
Whether you're looking for technology partners, engineering talent, or market insights — we can help connect you with the right Japanese organizations.
Get in Touch →